Dezincification has been an ongoing topic of conversation when discussing open loop copper plumbing systems. A prevailing misconception is that bronze valves are the only reliable option for dezincification resistance. However, due to changes in lead-free requirements as a result of the Safe Drinking Water Act, additional options have presented themselves as new, viable alternatives to bronze.
Heat treated, dezincification resistant brass valves are becoming a popular specified product within the engineering community. When subjected to International Organization for Standardization (ISO) 6509 testing, it is proven that heat treated dezincification resistant brass valves will perform as well as bronze in fighting against corrosion.
What is dezincification?
There are many forms of corrosion that can occur in copper based alloys. The most common in plumbing systems is dezincification. This process mainly exists in copper zinc alloys that contain more than 15% zinc. Dezincification selectively removes zinc from an alloy, leaving behind a porous and copper-rich structure with poor mechanical strength.
Additional environmental factors can also contribute to the acceleration of dezincification. These factors include water with high levels of oxygen and carbon dioxide, slow moving (stagnant) water, water with high chloride ion content and water with high salt content above room temperature.
In some instances, design engineers and contractors are unaware of harsh, preexisting conditions prior to beginning site-work. However, after a piping systems completion, there may be a number of visual signs indicating a higher likelihood of dezincification in common copper alloy valves. These indicators include, but are not limited to, mineral stains on the outer surface of a valve, water weeping from a valve body or stem seal and presence of a white deposit (zinc oxide) on the exterior of the valve.
A tightly written valve specification can help minimize these potential failures by simply calling out a proper corrosion resistant copper based alloy.
How can brass become corrosion-resistant?
There are two manufacturing methods that can eliminate dezincification in copper-based brass alloys. The first is to increase the copper content, which results in a reduction of zinc in the alloy. The second practice involves adding corrosion inhibitors to brass alloys. Only through a controlled heating and slow cooling annealing treatment can the second practice create a stronger material with a realigned molecular structure.
Brass alloys typically have two phases to their molecular structure. The first is alpha phase, which is dezincification resistant, and the second is beta phase, which is dezincification susceptible. When brass alloys are enhanced to an all alpha phase through heat treatment, they perform the same as bronze alloys when fighting dezincification.
The solution: Heat treated forged brass
An example of a heat treated alloy in the commercial plumbing industry is CW511L. This duplex brass is a lead-free brass alloy comprised of both an alpha and beta phase. Alpha and beta phases correlate to the solubility between metals in brass alloys, resulting in different colors and structures in each phase. As a result of the temperature changes in heat treatment, elements within the alloy change their liquid and solid states, resulting in a restructured alloy composition.
The alpha and beta composition is resistant to cracking and is extremely malleable at high temperatures. Figures 1.1 and 1.2 illustrate alloy reorganization during the heat treatment process. White circles represent the alpha phase, while black circles illustrate the beta phase.

Figures 1.1 and 1.2 illustrate alloy reorganization during the heat treatment process. Figures Courtesy of The Jomar Group
As mentioned previously, the purpose of heat treatment is to transform alpha and beta phases into a predominately alpha phase (dezincification resistant). After heat treatment, Figure 1.2 shows ‘sporadic beta phase’ being visibly represented as dark spots. The new composition structure of alpha to beta phase ratio is less than 1% beta phase, resulting in a dezincification resistant alloy.
At this point, beta phase is now referred to as a ‘residual beta phase’ and the alloy structure is now 99% in alpha phase. This leaves a brass alloy that is now truly dezincification resistant.
Figure 2 shows the varying phase changes of CW511L during the heat treatment process. These changes are distinguishable by the color and shape of the crystals within the alloy. When the alloy completes its heat treatment process, the color of the brass will appear more gold than bronze under a microscope. However, the valve body surface will appear bronze in color due to the oxidation of brass at 1,000° F (537.87° C).
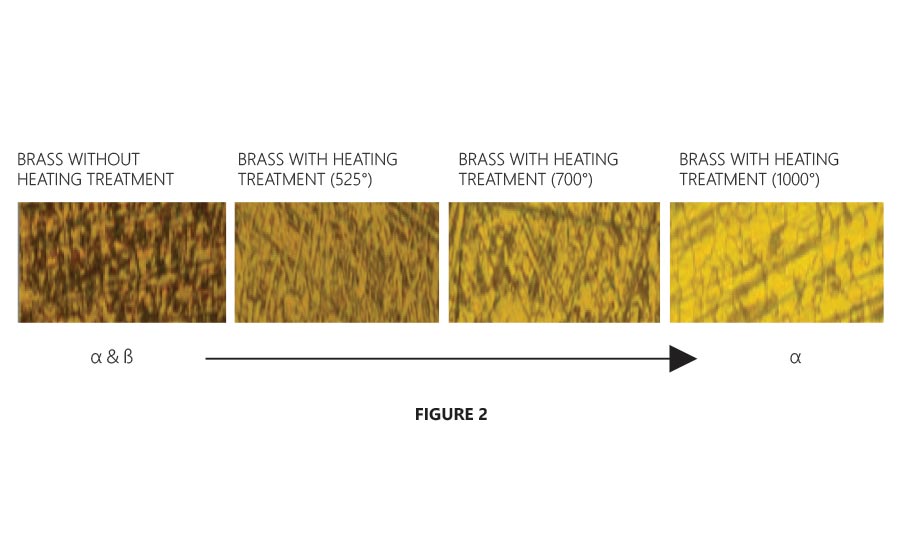
Figure 2 shows the varying phase changes of CW511L during the heat treatment process. Figure Courtesy of The Jomar Group
Figure 3 is the constitutional diagram of copper zinc alloys and depicts the effect of phase change based upon increased temperatures and the weighted percentage of zinc. When CW511L brass is heated to a temperature of 1,000° F (537.87° C), an all alpha phase is achieved. This alpha state is maintained in the alloy by a slow cooling, annealing process. The end result is optimal dezincification performance when tested to the ISO 6509 Standard.
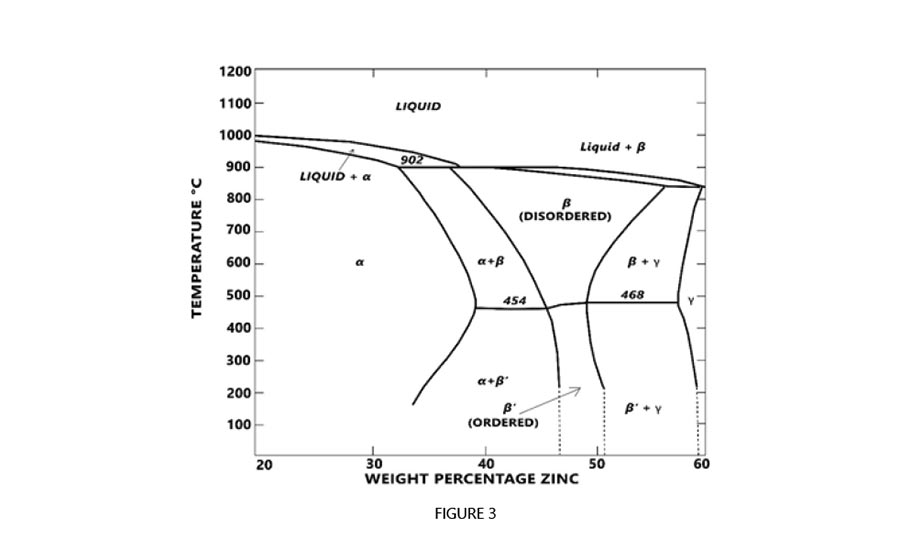
Figure 3 shows a constitutional diagram of copper zinc alloys and depicts the effect of phase change based upon increased temperatures and the weighted percentage of zinc. Figure Courtesy of The Jomar Group
Testing brass against the ISO 6509 Standard
ISO 6509 is the international testing standard for determining dezincification depth of copper alloys with zinc exposed to fresh, salin, or drinking water. The method is intended for copper alloys with a mass fraction of more than 15% zinc.
Third party ISO 6509 testing explored dezincification susceptibility amongst copper based alloys, including bronze and brass valves. This testing illustrated the difference in dezincification depths for each valve and corresponding alloy. It was determined that heat treated CW511L brass showed a dezincification depth of zero microns, which was equivalent to the dezincification depths seen in bronze valves. The remaining non-heat treated brass valves showed dezincification above 200 microns depth as illustrated in Figure 4.

Figure 4 shows the comparison of non-heat treated brass valves with heat treated CW511L brass. Figure Courtesy of The Jomar Group
This level of dezincification is above the acceptable performance level based on the ISO standard, deeming them as not truly dezincification resistant. The results of the test prove that not all brass materials are equal and that certain heat treated brass alloys can perform as well as bronze when it comes to corrosion resistance.
In closing, dezincification in plumbing systems is an ongoing battle, but bronze is not the only solution. New technologies, such as Jomar Valve’s heat treated CW511L brass alloy, can act as a combatant to dezincification while providing an increased level of mechanical strength.
Jomar’s 80 years of manufacturing experience has allowed the company to offer a cost-effective, engineered solution with its CW511L brass valves.



