
Issue: 9/03
Mad cow disease, West Nile virus, Anthrax, and now Monkey Pox, have all recently made news headlines. Consequently, many institutions and organizations are planning and building laboratories in order to secure research dollars to investigate dangerous biological agents. It is not uncommon for these facilities to require the use of laboratory animals for researching vaccines and other countermeasures. Laboratories that house large quantities of caged animals may require an automatic watering system to deliver the proper amount of this critical nutrient. Animals can go without food for days but will become stressed if deprived of water for even a few hours. Although the design professional will normally defer to the client concerning the decision of whether to use a watering system or not, it is important to review some of the factors involved in this process.
Approximately 90% of all animal research is conducted on rodents, due to their small size, preference to group housing and their genetic characteristics. Rodents can be housed by the thousands in what is referred to as ventilated cage racks (Figure 1). Maintaining these cage racks can place an enormous burden on laboratory technicians. The improvement in rack ventilation has allowed the frequency of cage cleaning to drop off dramatically and has made animal watering systems more prevalent. The alternative to automated watering systems is the use of water bottles, which requires an inordinate amount of the technician's time for changing bottles in order to maintain water quality.

Water Supply
Prepared feeds used in most facilities are low in moisture content, which means that animals need to drink more water to sustain their dietary requirement. Table 1 lists water requirements for some common laboratory animals.
Table 1. Average Daily Water Requirements
Animal Ounces/day
Rabbit 5.0
Guinea pig 3.0
Hamster 0.4
Gerbil 0.1
Rat 1.3
Mouse 0.2
Squirrel monkey 3.3
Rhesus monkey 30.0
Taken from AgSelect.com
The automatic watering system requires a clean water supply at an appropriate pressure, which is then carried through distribution piping to drinking valves at each cage position.
Whether the facility is fed from a municipal water supply or from a well, it may be prudent to consider how much drinking water to store in reserve in case of a disruption to the service. This disruption could be a result of equipment or distribution failure, or perhaps an extreme variation in water quality. Although installing a tank to provide a one- or two-day supply could reduce the severity of this problem, this approach should be thoroughly reviewed. A tank can also be used to administer medication or nutrients to animals, but it should be cautioned that the tank can facilitate the growth of water-borne pathogens.
If the control of water quality is deemed important, it will be necessary to consider a water treatment system. This will involve pre-filtration, to remove gross particles that can compromise the function of the watering system, and perhaps a water purification system such as reverse osmosis, which can remove up to 99.9% of most contaminants.
Of all the contaminants in the water supply, bacteria are the hardest to control and can live in purified water, even though it contains very few nutrients. The bacteria can go into a low-nutrient mode where they reduce in size and bond to the internal surfaces of watering systems. Bacteria will attach to any surface that water contacts and develop what is termed a biofilm. Biofilm development can occur in animal watering system piping, and as the biofilm grows, pieces of it can detach and flow downstream to be consumed by the animals. To control free-floating bacteria, it is important to treat the water with a residual disinfectant. When combined with automated flushing, low-residual chlorination or acidification is effective in controlling bacteria.
Water Purification
Reverse osmosis(RO) is a water purification process in which water is forced by pressure through a semi-permeable membrane. In normal osmosis, water flows from a less concentrated solution through a semi-permeable membrane to a more concentrated solution. Reverse osmosis uses pressure to reverse normal osmotic flow. Water flows from a more concentrated solution through a semi-permeable membrane to a less concentrated solution. The feedwater to the reverse osmosis system flows over the surface of the membrane, and a percentage of the water is forced through by pressure and becomes the purified water or permeate. The remaining water, the concentrate, retains the rejected contaminants and is drained off. The percentage of feedwater that is recovered as permeate, called percent recovery, is typically 33% or 50%. The other 66% or 50% is discharged to drain.
Other water purification techniques include distillation, which will remove a wide range of contaminants through the boiling of feedwater and collecting the resulting condensate. This process is more energy-intensive and is consequently more expensive to operate than reverse osmosis systems. Scale formation can also be a problem with distillation units. Deionized or DI water is purified by passing water through ion-exchange resins which remove dissolved ionized chemicals. However, deionization does not remove organic chemicals, bacteria or other microorganisms. Colonies of microorganisms can become established and proliferate on the nutrient-rich surfaces of the resin. If not regularly sanitized or regenerated, ion-exchange resins themselves can contaminate drinking water with bacteria. Ultrapure water contains very low amounts of chemical impurities and has a very low electrical resistance. In the laboratory, ultrapure water usually refers to Type 1 reagent grade water. RO drinking water is not ultrapure, as the process removes approximately 93-97% of dissolved ionized chemicals from the feedwater, which means that 3-7% of minerals pass through the membrane into the drinking water. The purity of ultrapure water is about 100 or more times greater than RO drinking water. Ultrapure water is more aggressive than RO water and requires more capital and operating expenditures than an RO system. An ultrapure water system will typically start with RO-quality water and purify it further with distillation or deionization.
Distribution System
Purified water is an aggressive solvent that will dissolve certain materials it comes in contact with, which can lead to water quality degradation. To maintain the purity of the water until it reaches the animal, the following automated watering system design features should be incorporated.Wetted materials should be 316 stainless steel, though plastics are acceptable for storage and also piping materials for RO water. Copper and brass components should not be used. Piping should be laid out to ensure flow through the entire system. Stagnant dead-end lengths of pipe should be avoided. Serpentine distribution piping increases the effectiveness of flushing procedures. Flushing systems should be employed, as the flow of water through an automated watering system during normal use is low and intermittent. Regular high-pressure flushing will exchange the water and help control bacteria. Depending on the size and complexity of the system, either manual or automated flushing systems can be employed for room distribution system and rack manifold piping. Constant water recirculation is another method of reducing water stagnation. Water would flow from a storage tank, through an ultraviolet disinfection unit, to the room distribution system, and back to the tank.
A reduced-pressure-principle backflow device (RPBD) should be installed in the water supply ahead of the water purification system to guard against back-siphonage to other parts of the facility. After passing through the water treatment equipment, the water arrives at the storage tank, where it is pumped to the animal holding rooms through a main distribution header. Before entering the animal rooms, the drinking water passes through a pressure-reducing station, where the pressure is reduced from the 40 to 60 psig range down to approximately 3 to 5 psig. Where automated flushing is employed, the water is first regulated down from house pressure to 15 psig as it goes through a high-pressure regulator before entering a low-pressure regulator taking the water pressure to 3 psig. This is so that the flushing cycle can be operated at the higher pressure of 15 psig by simply bypassing the low-pressure regulator. The pressure-reducing station should contain pressure sensors to monitor and detect problems with pressure and flow. The sensors should give an audible and visual alarm to alert staff of any problems.
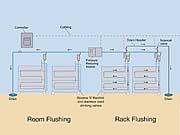
Manifolds
At this point, the piping enters the animal holding room and runs along the walls. The pipe should be mounted above rack height so that it won't be damaged inadvertently. In rodent rooms, the room distribution piping should be connected to the mobile racks with a recoil hose, allowing the watering system to be disconnected so that racks can be moved to different rooms or for washing.
A manifold is the piping that is mounted on each rack, cage or kennel. Connected to the manifold at each cage position are drinking valves through which the animals access water. Manifolds should be designed specifically for each manufacturer's racks. For animals housed in racks, the piping should be routed in what is referred to as a reverse "S" design (Figure 2), which allows air to be purged from the manifold. This configuration also provides a continuous section of pipe with no dead legs, allowing the piping to be completely flushed.
Automatic Flushing
A central animal watering system controller can be used to automate the flushing of rack manifolds and room distribution piping to avoid stagnation and to combat bacteria growth. The controller can be programmed to automate pan and trench waste flushing as well. Flushing is initiated by a transition to higher water pressure and the subsequent actuation of solenoids installed at piping termination points. This high-pressure flush, usually 15 psig, provides a more vigorous and more complete flush of the manifold than if it were done at the normal operating range of 3 to 5 psig. After a few minutes, the pressure is indexed to normal, and the solenoid valve closes. The water is flushed from the manifolds through the drain lines to a nearby sink or drain. This flushing process enhances water quality by bringing fresh water into the piping.
The controller should be specified to simultaneously monitor the watering system, verify and document the flush sequences and check for high and low pressure and irregular flow conditions. If problems occur, the controller can be programmed to send an alarm to notify facility staff. Flushing and alarm events can be recorded and log reports can be generated to facilitate program validation procedures.


